In today’s competitive clinical research landscape, Clinical Research Organizations (CROs) are under immense pressure to deliver faster trials, greater efficiency, and regulatory compliance—all while managing complex sponsor expectations. Traditional tools like spreadsheets or outdated CTMS platforms often fall short. That’s where a modern Clinical Trial Management System (CTMS) becomes a game-changer.
In this article, we’ll explore why CROs need modern CTMS solutions and highlight the top 10 benefits driving adoption across the clinical research industry.
1. Simplified Trial Operations
A modern CTMS centralizes everything—from study startup to closeout. CROs no longer juggle multiple disconnected tools. Instead, they gain a single source of truth, making trial management more efficient and less error-prone.
2. Faster Study Start-Up
Delays in startup cost sponsors both time and money. With modern CTMS, CROs can automate site selection, feasibility, and document workflows, drastically reducing startup timelines and boosting client satisfaction.
3. Enhanced Regulatory Compliance
Regulatory demands from DCGI, FDA, EMA, and ICH-GCP are more stringent than ever. A modern CTMS ensures audit-ready documentation, version control, and electronic approvals, helping CROs stay compliant and inspection-ready at all times.
4. Improved Sponsor Collaboration
Sponsors expect real-time visibility into study progress. With modern CTMS dashboards and reporting, CROs can provide sponsors with transparent updates, strengthening trust and ensuring long-term partnerships.
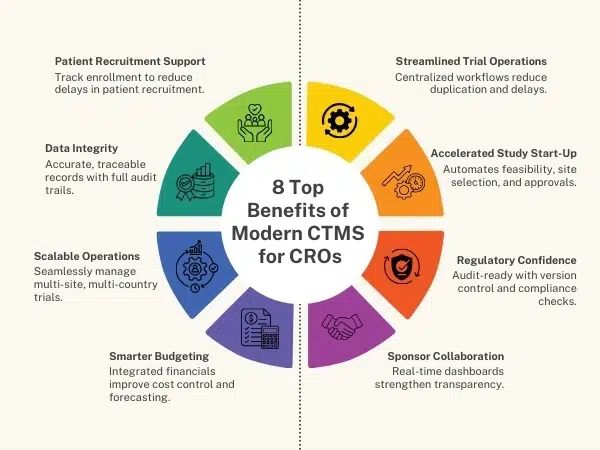
5. Optimized Resource & Budget Management
A modern CTMS integrates budget tracking, payments, and resource allocation, allowing CROs to forecast costs accurately and prevent budget overruns. This not only improves margins but also enhances competitiveness in bids.
6. Scalable Across Multiple Trials
CROs often handle multiple studies across geographies. A modern CTMS supports multi-site, multi-country, and multi-protocol management, enabling CROs to scale without compromising on quality or timelines.
7. Enhanced Data Accuracy & Traceability
Manual data handling leads to errors and compliance risks. Modern CTMS platforms ensure data integrity, audit trails, and role-based access control, reducing risks while maintaining transparency for sponsors and regulators.
8. Boosted Patient Recruitment & Retention in Clinical Trials
Patient recruitment is the biggest bottleneck in clinical trials. With integrated recruitment modules, CTMS helps CROs track site performance, enrollment rates, and patient engagement, ensuring studies stay on schedule.
Final Thoughts
For CROs, the shift from legacy systems to modern CTMS is no longer optional—it’s a strategic necessity. From accelerating trial timelines to ensuring compliance and boosting sponsor confidence, modern CTMS platforms like ImproWise empower CROs to deliver trials with speed, accuracy, and efficiency.
👉 If you’re a CRO looking to streamline operations and win more sponsor partnerships, now is the time to adopt a modern CTMS.


